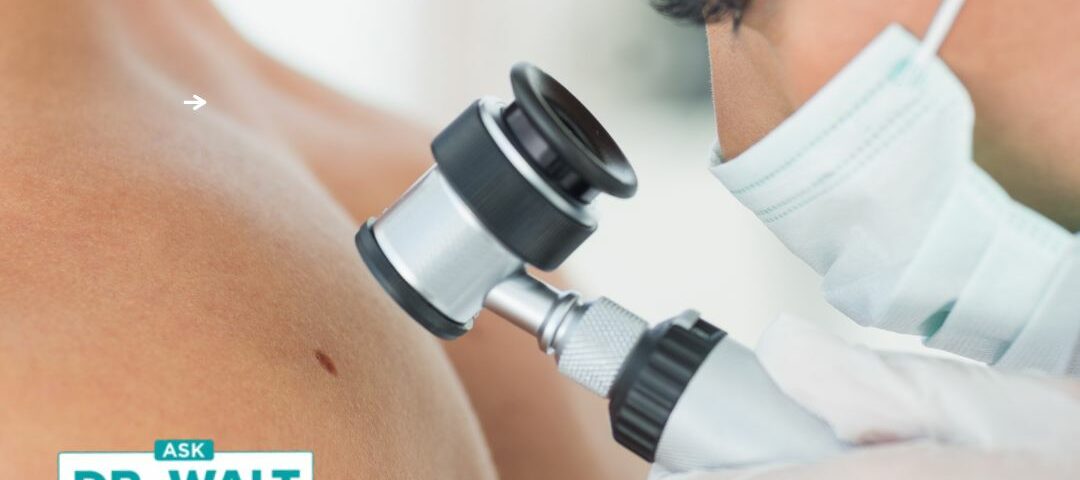
FDA issues warnings about products to remove moles and skin tags

Sunday Note – Post Abortion Ministry
August 14, 2022
Tuesday Patient Stories – Kissimmee Tales Chapter 1B – Near Death
August 16, 2022Healio reports, “The FDA has issued warnings to three companies for selling unapproved products to remove moles or skin tags, according to a press release.”
The warning letters were sent to companies, all of which were said “to be in violation of the Federal Food, Drug, and Cosmetic Act by selling products that claim to remove moles or skin tags.”
The FDA added, “Moles should be evaluated by a health care practitioner. Self-diagnosis and treatment of moles could lead to delayed cancer diagnosis and treatment, and even cancer progression.”
The FDA has also issued a consumer warning noting that products marketed for removing moles and other skin lesions can cause injuries and scarring.
The sale of these products risks public health and may jeopardize consumers’ health when used without consulting a health care professional.
The FDA issued the warning letters to:
HealthDay reports that “no over-the-counter medications have FDA approval for that purpose.”
© Copyright WLL, INC. 2022. This blog provides healthcare tips and advice that you can trust about a wide variety of general health information only and is not intended to be a substitute for professional medical advice, diagnosis, or treatment from your regular physician. If you are concerned about your health, take what you learn from this blog and meet with your personal doctor to discuss your concerns.